Зачем вам участвовать в клиническом испытании?
4 мин. чтения

Больной раком с множественной миеломой рассказывает, почему он решил принять участие в клиническом испытании
Несомненно, лечение рака улучшилось в разы, и новые достижения происходят постоянно. При наличии многочисленных перспективных методов лечения - не говоря уже о порой непосильных логистических деталях, связанных с координацией лечения рака, - зачем вам участвовать в клинических испытаниях? Ламар Норьега, пациент Онкологических центров Rocky Mountain (RMCC), рассказывает, что побуждает его участвовать в клинических испытаниях.
У Ламара Норьеги не было никаких симптомов рака, когда в мае 2017 года в возрасте 57 лет ему был поставлен диагноз. Это не является необычным для того типа рака, который его лечащий врач обнаружил по анализу крови во время обычного медосмотра: множественная миелома.
Через неделю Ламар поступил в RMCC, чтобы встретиться со своей командой онкологов, включая доктора Резника и доктора Берка.
"Множественная миелома имеет 3 стадии", - сказал Ламар. На той стадии, которая была у меня, она "тлела", но не была активной. Иногда первая стадия может длиться 20 лет - когда она есть, но ничего не делает. Мои показатели были очень хорошими. Доктор Берк сказал, что с учетом моего здоровья, возраста и ранней стадии диагноза. Я был идеальным кандидатом для клинических испытаний".
Клинические испытания помогают будущим пациентам
Участие в клинических испытаниях означает, что результаты и опыт пациента используются для изучения получаемого им лечения с целью повышения эффективности и снижения рисков для будущих пациентов. Для Ламара это было важным фактором при выборе участия в клинических испытаниях.
"Мы потеряли мою мать из-за рака", - сказал он. Он также потерял несколько других родственников, заболевших раком. "Я подумал, что если это поможет мне и поможет другим на этом пути, то оно того стоит".
Клинические испытания организуются по-разному в зависимости от изучаемого лечения и могут длиться в течение различных периодов времени. Например, некоторые из них имеют ограниченный период времени, а некоторые продолжают сбор данных от пациентов в течение всей их жизни.
Ламар решил принять участие в двух различных клинических испытаниях. Одно из них, регистрационное исследование The Circulating Cell-free Genome Atlas (CCGA) Study (GRAIL-001), призвано помочь врачам лучше понять, почему у одних людей развивается рак, а у других нет, путем изучения их генов и диагноза рака в течение пяти лет. Хотя участие в исследовании может дать врачам некоторые сведения о генах Ламара и сравнить его с другими пациентами, похожими на него, его участие, скорее всего, не окажет прямого влияния на лечение рака. Однако Ламару приятно осознавать, что он помогает ученым лучше понять его болезнь.
Клинические испытания дают шанс воспользоваться последними достижениями в лечении рака
Второе испытание, в котором участвовал Ламар, A Study to Evaluate Dara-CyBorD in Previously Untreated and Relapsed Subjects With Multiple Myeloma, позволило Ламару воспользоваться перспективными препаратами, еще не утвержденными для общего использования, и во многом определило курс лечения Ламара.
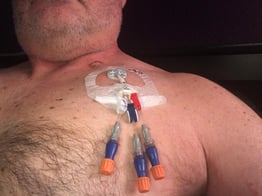
"Первый курс лечения я должен был пройти в течение двух дней, чтобы убедиться, что у меня нет побочных реакций на препарат. Затем его стали вводить раз в неделю", - говорит Ламар. "Мне поставили порт, что значительно облегчило жизнь. Поскольку я хорошо реагировал на химические препараты и был в хорошей форме, в феврале 2018 года мне сделали пересадку стволовых клеток, что дало бы больше шансов на хорошее здоровье в долгосрочной перспективе."
Врачи приостановили еженедельные инфузии химиотерапии Ламара на два месяца, чтобы сделать пересадку стволовых клеток и дать его организму время на восстановление. Затем он вернулся к клиническим испытаниям.
Сейчас испытания завершены, и лекарство одобрено для применения у других пациентов, таких как Ламар. Однако лекарства оказались настолько эффективными для Ламара, а его организм хорошо переносил лечение, что он продолжает получать их для поддержания ремиссии рака.
"Я буду принимать поддерживающий препарат до тех пор, пока он работает", - сказал Ламар. "В конце концов, препарат перестанет действовать, но мы сможем перейти на другой одобренный препарат или провести еще одно клиническое испытание (для лекарства, еще не одобренного FDA)".
В целом, Ламар остался более чем доволен своим лечением рака и решением участвовать в клиническом испытании.
"Мне очень повезло. У меня ничего не случилось, стучу по дереву", - сказал он.
Недавно он даже смог переехать - из Колорадо в Миннесоту - без какого-либо перерыва в лечении рака, которое будет продолжаться до конца его жизни. Сейчас Ламар живет к западу от Сент-Пола, чтобы быть ближе к семье, и сказал: "Перенос лечения прошел очень гладко. Доктор Берк - своего рода "главный специалист по множественной миеломе", и он направил меня к доктору в Миннесоте (который был частью центра RMCC). Онкологическая сеть США, как и RMCC). Вчера я прошел первый курс лечения".
Выбор участия в клиническом исследовании может быть очень личным решением, основанным на ваших приоритетах и уникальной ситуации. Если вы задаетесь вопросом, почему вы должны участвовать в клинических испытаниях, вам может помочь чтение историй о том, что побудило других пациентов принять участие в клинических испытаниях, таких как Джеки Тернер, Вивиан Барфути Деб.